Pharmaceuticals and Biotech
-
Home
-
Pharmaceuticals and Biotech


Key Findings from the 2025 Global Compliance Survey
Our survey of 120 QA, QC, and risk executives across pharma, biotech, and CDMOs reveals:
- 72% cite data integrity gaps as the #1 inspection risk. Paper-hybrid systems and inconsistent audit trails remain widespread.
- 65% highlight uncertainty in applying AI/ML under GxP. Most firms lack structured validation or monitoring frameworks.
- 59% of CDMOs report that client audit frequency has increased by >25% since 2020. Audit scope is also broader, covering digital and vendor systems.
- 41% of respondents still use paper batch records in at least one facility, despite regulators' urging for digitisation.
- Seventy-four per cent believe digital audit readiness will become a board-level mandate within two years.
- Only 18% run a validated AI governance framework (GMLP) — exposing them to risk of unexplainable system outputs.
Why Act Now
- Rising enforcement: FDA warning letters increased 25% from 2020–2024, with data integrity cited in >60% of cases [FDA, 2024].
- Regulatory tightening: EMA and CDSCO have both issued new expectations on AI/ML transparency and digital records.
- Economic stakes: Cost of a single consent decree can exceed $500M (public cases), while digital-first programs often yield 2x ROI through avoided penalties and efficiency gains.
Bottom line: Leaders who invest now can position compliance as both a risk shield and growth enabler.
Drivers of Change
-
Complex global supply chains
-
Faster speed-to-market demands
-
Data volumes exploding
-
Rising regulatory scrutiny
-
AI adoption pressure

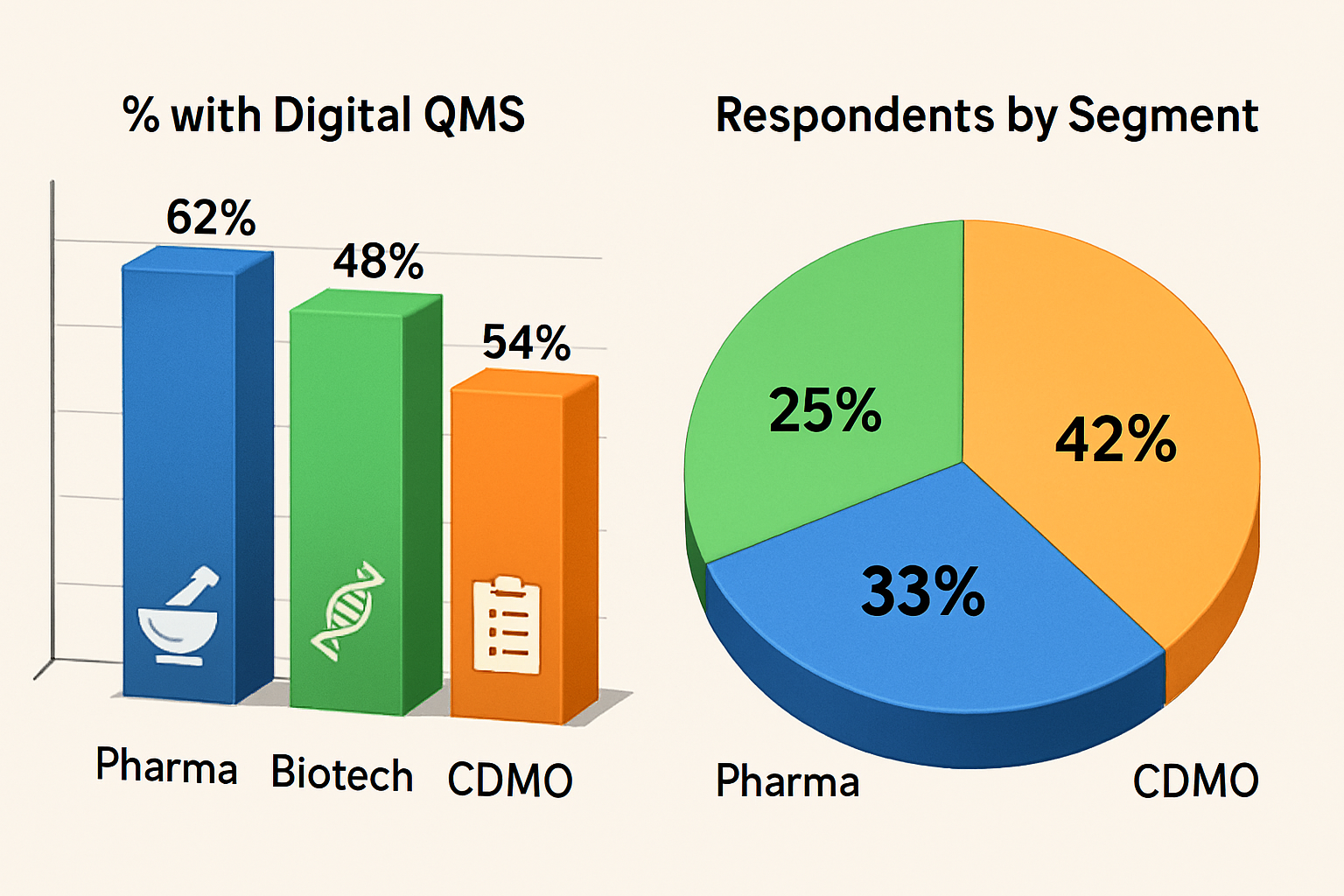
Survey Methodology & Respondent Profile
-
Pharma Segment Priorities
-
Biotech Segment Challenges
-
CDMO Segment Needs
-
Digital QMS Adoption Trends
Key Regulatory Frameworks
- FDA 21 CFR Part 11 (Electronic Records & Signatures): Foundational requirement for trust in digital records.
- EU Annexe 11 (EMA, Computerised Systems): Emphasis on validation, security, and audit trails.
- ICH Q9 (R1, effective July 2023): Enhanced risk management, with explicit mention of digital systems.
- CSA (Computer Software Assurance, FDA draft 2024): Shifts validation to a risk-based approach — focus on intended use, not documentation volume.
CDSCO India (2023 Guidance): Push for electronic batch records and audit trails in manufacturing sites.
Recent Regulatory Announcements (Timeline)
| Year | Authority | Update | Key Impact |
|---|---|---|---|
| 2021 | FDA | AI/ML Action Plan | Framework for adaptive AI; regulators signal future GMLP. |
| 2022 | EMA | Reflection Paper on AI in Medicine | Encourages explainability, reproducibility, oversight. |
| 2023 | ICH | Q9(R1) effective | Clearer linkage between risk management and digital oversight. |
| 2023 | CDSCO | Data Integrity & e-Records Circular | Requires validated audit trails in manufacturing. |
| 2024 | FDA | Draft on CSA (Computer Software Assurance) | Encourages risk-based validation; less documentation burden. |
Business Challenges in Achieving Digital-First Compliance
Our 2025 survey and interviews reveal six recurring barriers that slow digital-first, audit-ready compliance adoption across pharma, biotech, and CDMOs
- Fragmented Legacy Systems
- Validation Burden (CSV/CSA Transition)
- Resource & Capability Constraints
- Vendor Oversight & Cloud Qualification
- AI/ML Governance Ambiguity
- Change Resistance & Culture

Case Study for Large Pharma
Context & Pain Points
-
Top-10 pharma, 50+ manufacturing sites worldwide.
-
3 FDA 483s in 2 years citing data integrity & audit trail gaps.
-
Hybrid paper-digital processes; partial LIMS, manual batch release.
-
Rising risk after competitor’s $600M consent decree.

Solution Implemented
Digital-first compliance transformation led by a cross-functional task force.
- Consolidated 8 legacy LIMS systems
- Adopted Computer Software Assurance
- Established central AI Governance Board
- Deployed a real-time compliance dashboard
Timeline & Maturity
- Year 1: Pilots in 2 sites, eQMS roll-out, CSA training.
- Year 2: Global LIMS consolidation, compliance dashboard deployed.
Year 3: AI-assisted QC anomaly detection system validated under GMLP principles.
| KPI | Before | After | % Improvement |
|---|---|---|---|
| FDA audit observations per site (avg) | 7 | 2 | -71% |
| Time-to-batch release | 12 days | 8 days | -33% |
| % of systems validated under CSA | 0% | 80% | N/A |
| Audit readiness confidence (self-rated) | 58% | 92% | +34 pts |
Case Study for Mid-size CDMO
Context & Pain Points
-
CDMO Scale
-
Revenue Risk
-
Operational Gaps
-
Client Demands

Timeline & Maturity
Months 1–6: eQMS go-live, training completed.
Months 6–12: AI deviation pilot in QC.
Year 2: Expansion to document management and training records.
Measured Outcomes
| KPI | Before | After | % Improvement |
|---|---|---|---|
| Average deviation closure time | 45 days | 12 days | -73% |
| Trial audit findings (per inspection) | 5 | 1 | -80% |
| Compliance staff FTEs needed | 6 | 4 | -33% |
| Investigator queries on audit trail | Frequent | Rare | Major reduction |
Share this service:
Leave a comment
Your email address will not be published. Required fields are marked *
